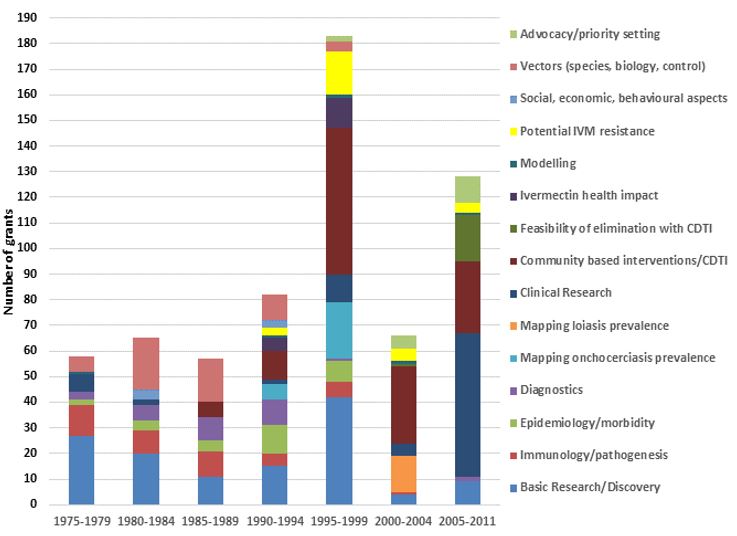
Research area | Most significant outputs | Most significant outcomes |
| Basic research/discovery | The establishment of in vitro and in vivo screening systems allowed the identification of ivermectin and moxidectin as potential anti-onchocercal drugs
Efficacy of emodepside against O. volvulus macrofilariae ex vivo. | Initiation of clinical development of ivermectin (by Merck) and moxidectin (by TDR)
Regulatory registration of ivermectin (without TDR support) and of moxidectin (with TDR support)
Large scale studies to evaluate ivermectin and moxidectin efficacy and safety during Mass Drug Administration
Initiation of further evaluation of emodepside by the Drugs for Neglected Diseases Initiative (DNDi) as a potential anti-onchocercal drug |
| Diagnostics | DNA probes to distinguish forest and savanna strains of O. volvulus and of O. volvulus and O. ochengi | Development of O-150 based pool screening, the method used for determining vector infectivity – the criterion for stopping interventions in WHO guidelines |
| Epidemiology/morbidity | Identification of the health and socioeconomic burden of skin disease in Central and Eastern Africa | Formation of the African Programme for Onchocerciasis Control (APOC) |
| Mapping onchocerciasis prevalence | Method for rapid epidemiological assessment and mapping of onchocerciasis prevalence | Identification of APOC areas for implementation of Community directed treatment with ivermectin (CDTI) |
| Mapping loiasis prevalence | Method for rapid epidemiological assessment and mapping of loiasis prevalence
Correlation of Loa loa prevalence and microfilaraemia | Mapping of loiasis co-endemic areas where CDTI can only be rolled out only with a special strategy to minimize sequaelae of severe and/or serious adverse reactions |
| Clinical research | Network of onchocerciasis researchers facilitating Merck’s ability to develop a collaborative relationship for ivermectin clinical evaluation
Methods for quantifying clinical reactions to microfilaricides
Efficacy and safety data from phase II and III studies of moxidectin
Data on the dependency of adverse reactions to ivermectin in Loa loa infected individuals on pre-treatment microfilaraemia
Identification of individuals with lower than expected response to ivermectin (suboptimal responders) | Data on safety and efficacy of ivermectin supporting submissions for regulatory registration
Data on safety and efficacy of moxidectin supporting submissions for regulatory registration
Strategy for implementation of CDTI in onchocerciasis endemic areas depending on Loa loa prevalence.
Further studies (not TDR funded) into the prevalence of suboptimal responders in areas with long term CDTI in Ghana and Cameroon. |
| Community based interventions/ CDTI | Safety of ivermectin during large scale use.
Feasibility and effectiveness of community-directed treatment
Ivermectin dosing by weight compared to height | Community directed treatment with ivermectin (CDTI) as the current standard strategy for onchocerciasis control and elimination
Ivermectin dosing during CDTI based on height rather than weight |
| Feasibility of elimination with CDTI | Data showing that biannual or annual CDTI can reduce vector infectivity to the level WHO guidelines require for stopping CDTI | Change in APOC objectives from the control of onchocerciasis as a public health problem to the elimination of parasite transmission where feasible. |
| Ivermectin health impact | Effect of ivermectin treatment on eye and skin disease | Formation of the African Programme for Onchocerciasis Control (APOC) |
| Modelling | Enhancement of the transmission model ONCHOSIM with a module for adult form fecundity and the impact of drugs on fecundity | Contribution to the utility of ONCHOSIM for estimating the impact of drugs with effect on adult form fecundity (such as ivermectin) for estimating the effect of drug treatment on infection prevalence and intensity. |
| Vectors (species, biology, control) | Identification of Bacillus thuringiensis H-14 as a safe and effective biological insecticide with several commercial preparations of Bacillus thuringiensis H-14 registered
Feasibility of vector elimination in Bioko Island | B.t. H-14 use by OCP for vector control from the time temephos resistance developed
Additional insecticide for rotational larviciding to minimize the development of insecticide resistance
APOC funded vector elimination in Bioko Island |